UNIST site map
- Admissions
-
Academics
- Colleges and Schools
-
Academic Affairs
- Academic Calendar
- Academic Curriculum
- Requirements for Graduation
- Browse Open Courses
- Undergraduate Administration
-
Graduate Academic Affairs
- Tuition Fee Payment
- Academic Leave of Absence/ Academic Return
- Voluntary Withdrawal/ Expulsion
- Change of Major
- Change of Degree Program/ Dropping of Degree Program
- Class Period/ Attendance Period/ Academic Year・Semester
- Course Registration
- Course Drop
- Attendance/ Grade/ Exam
- Credit Transfer/ Credit Carryover
- Academic Forms
- Education Support
-
Research/Industry
- Research Aims
- Research Findings
- Researcher Search
-
Research Organizations
- UNIST Multi-Interdisciplinary Institute
- IBS Research Groups
-
UNIST Labs
- Department of Mechanical Engineering
- School of Energy and Chemical Engineering
- Department of Civil
- Department of Materials Science and Engineering
- Department of Nuclear Engineering
- Department of Industrial Engineering
- Department of Design
- Department of Biomedical Engineering
- Department of Biological Sciences
- Department of Electrical Engineering
- Department of Computer Science and Engineering
- Department of Mathematical Sciences
- Department of Chemistry
- Department of Physics
- School of Business Administration
- Graduate School of Carbon Neutrality
- Graduate School of Artificial Intelligence
- Research Support
- University-Industry Relations
- Campus Life
- News Center
- About UNIST
-
etc
- UNIST Bulletin
- Work-Life Balance Support System
- UNIST Gender Equality Plan
- Faculty Invitation for Tenure Track
- Faculty Invitation for Non-Tenure Track
- Board Meeting Minutes
- University Council Meeting Minutes
- Administrative Service Charter
- Privacy Policy
- Copyright Policy
- Rejection of Unauthorized Email Collection
- Information Disclosure


Connection Points of Knowledge, Everything About UNIST
Try searching.
Recommended search terms


- portal
- U Academics Innovation Center
- Leadership Center
- Dormitory
- Academic Information Center
- International Students Support
- Browse Open Courses
- Course Registration
- Graduation Requirements for Graduation
- Academic Leave of Absence/ Academic Return
- Military Service
- Certificate Issuance
- Academic Calendar
- Scholarships
- Campus Map
- Campus Life Guidebook
- Health Care Center
- Human Rights Center
- portal
- Job Opening
- Announcement for Bid
- UNIST AI Services
- UNIST Daycare Center
- Sports Center
- UI Downloads
- Announcement
- Recruitment of Professors (Non-tenure)
- Faculty Invitation for Tenure Track
- UNIST Academic Information Center
- Office of Research Facilities and Training
- Office of Research Affairs
- Rule Management System
- Academic Calendar


NEWS CENTER
Discover not only Research Findings and event news, but also the diverse facets of UNIST presented by reporters and writers.
News Center
UNIST News
Turning Solar Panel Waste into Hydrogen and High-Value Silica
The research was published in the online version of Joule on March 27 and highlighted in its Future Energy section.
- Research
- JooHyeon Heo
- 2026.04.14
- 349
Summary
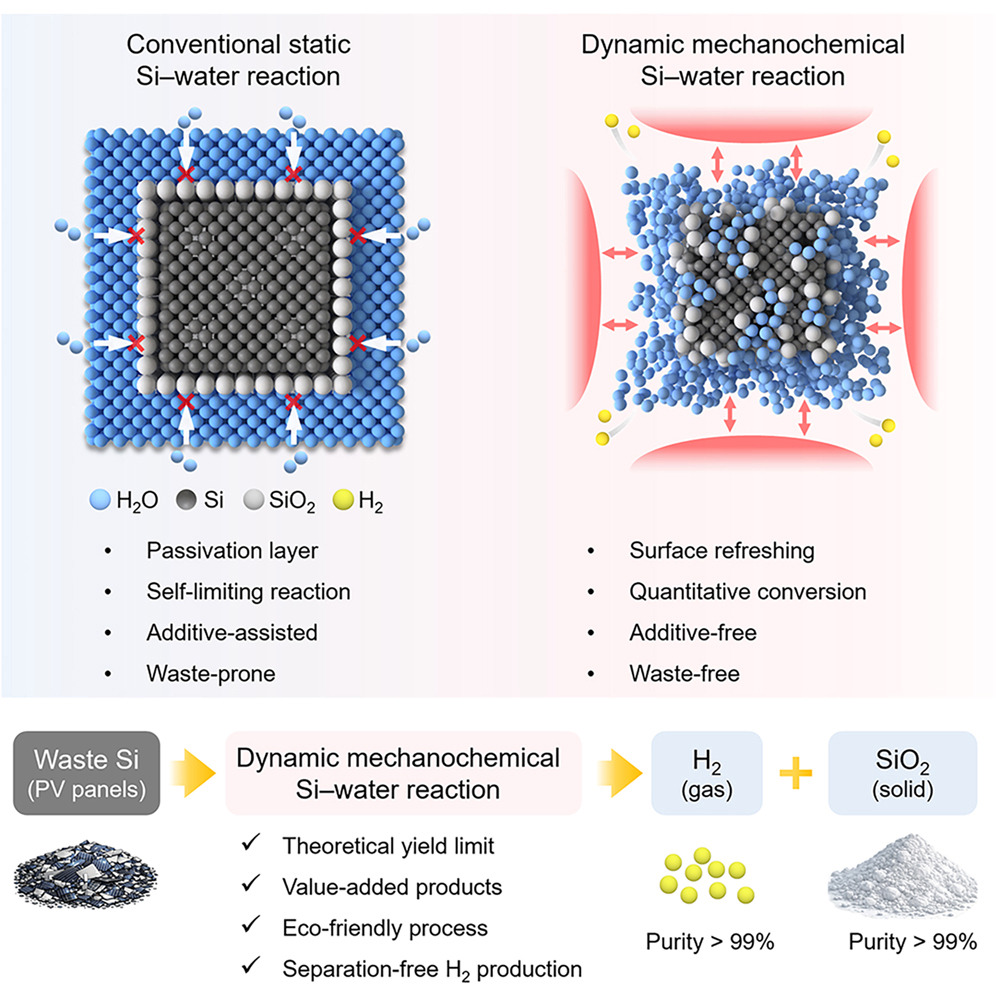
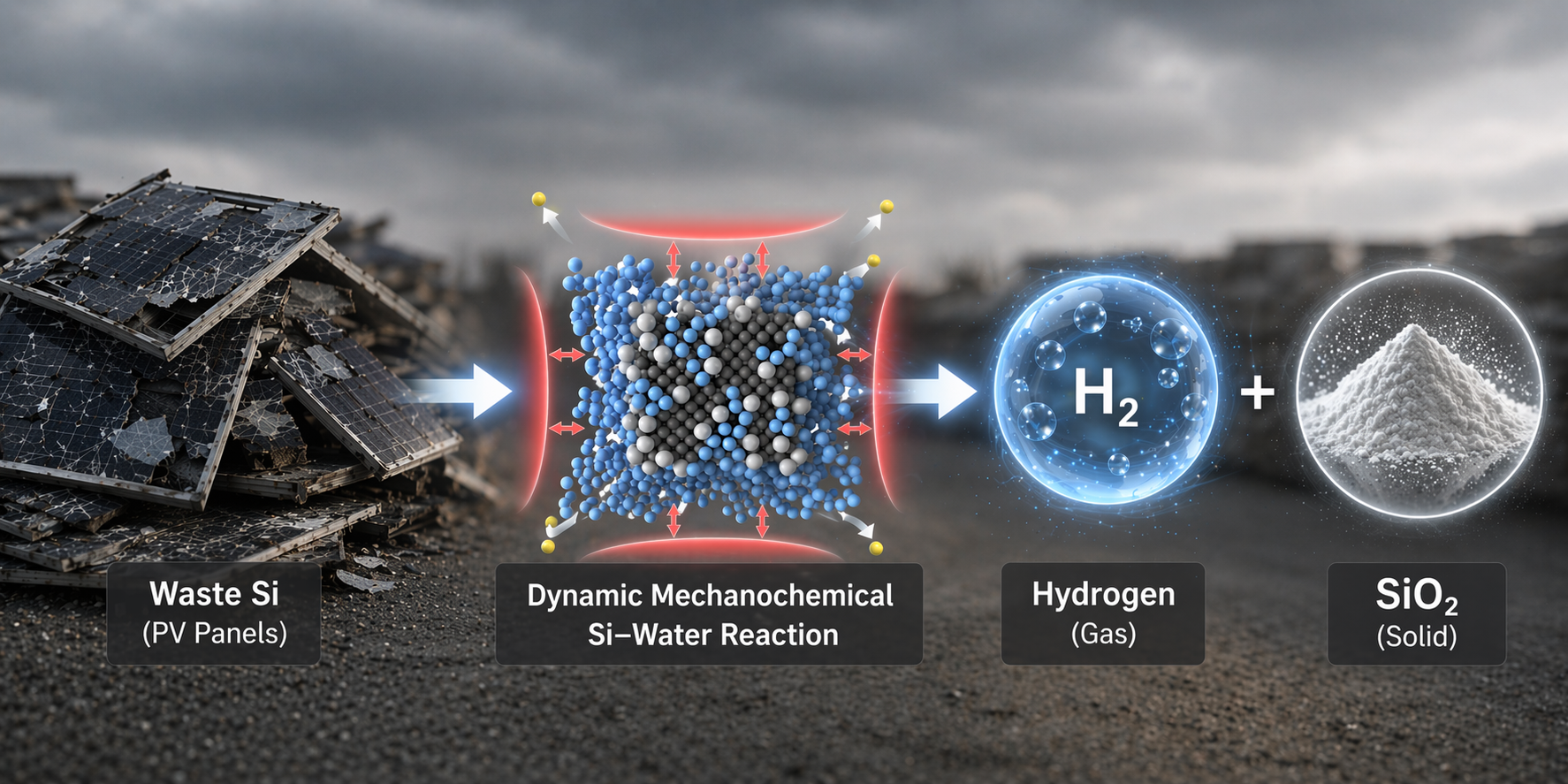
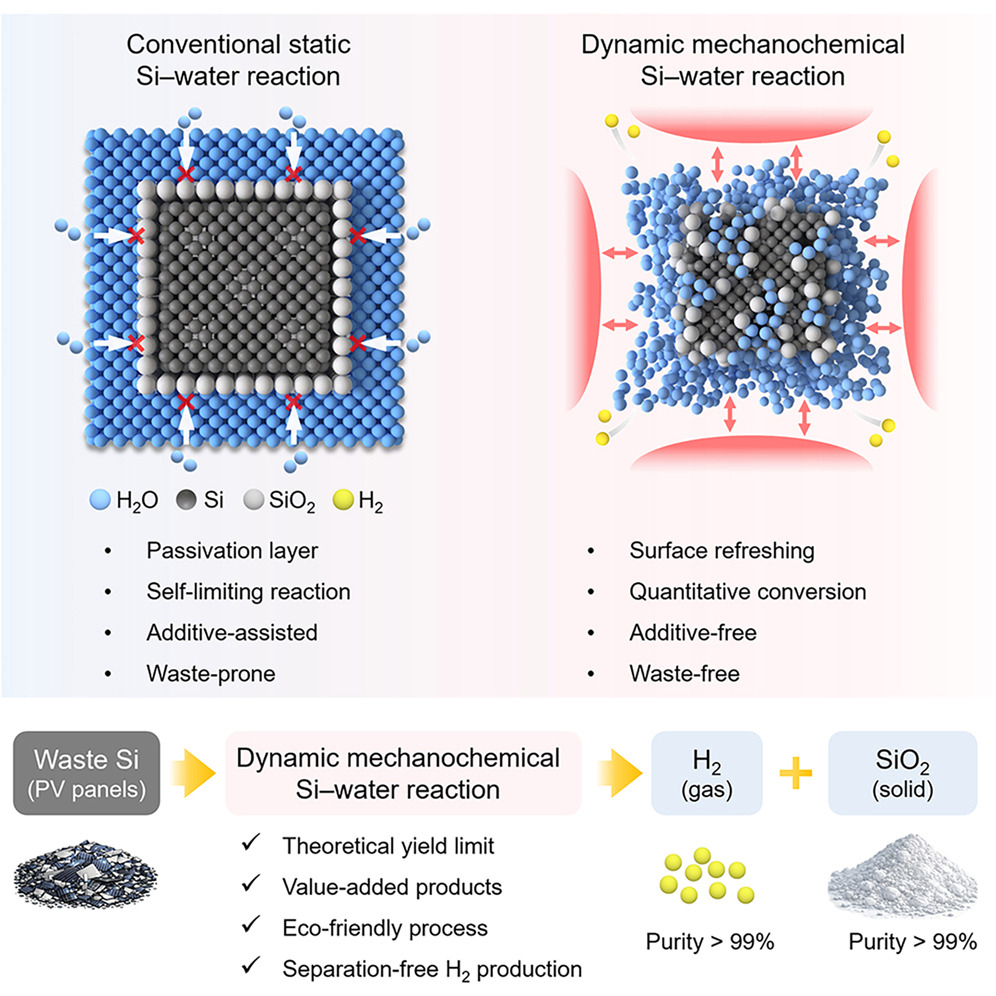
In response to the growing accumulation of end-of-life solar panels, researchers at UNIST have unveiled an innovative, environmentally friendly method to convert photovoltaic silicon into high-purity hydrogen and valuable silica. Led by Professor Jong-Beom Baek from the School of Energy and Chemical Engineering at UNIST, this breakthrough promises to revolutionize solar panel recycling and sustainable hydrogen production.
The team led by Professor Jong-Beom Baek developed a mechanochemical process that overcomes the self-limiting silica passivation layer on silicon surfaces. By placing silicon and water with small abrasive beads into a rotating vessel, repeated mechanical collisions strip the silica layer, enabling the reaction to proceed to nearly its theoretical maximum. Experimental results show approximately 1,706 mL of hydrogen per gram of silicon—achieving 99.6% of the maximum yield, significantly surpassing conventional thermochemical methods.
Moreover, the silica byproduct serves as an effective catalyst support. When used with nickel catalysts, it enhances carbon dioxide conversion and methane selectivity, thanks to its high surface hydroxyl density that improves catalyst dispersion.
“By leveraging waste silicon from decommissioned solar panels, our process produces high-purity hydrogen efficiently while also recovering valuable silica for industrial applications,” says Professor Jong-Beom Baek. “This approach not only advances sustainable energy but also contributes to resource circularity and environmental protection.”
This technology offers a cost-effective, scalable, and environmentally benign alternative to traditional photovoltaic waste management. Operating continuously, the process boasts higher productivity and energy efficiency, making it suitable for industrial deployment. It paves the way for a circular economy in solar energy, transforming waste into valuable resources and supporting the global shift toward clean hydrogen.
Related Links