UNIST site map
- Admissions
-
Academics
- Colleges and Schools
-
Academic Affairs
- Academic Calendar
- Academic Curriculum
- Requirements for Graduation
- Browse Open Courses
- Undergraduate Administration
-
Graduate Academic Affairs
- Tuition Fee Payment
- Academic Leave of Absence/ Academic Return
- Voluntary Withdrawal/ Expulsion
- Change of Major
- Change of Degree Program/ Dropping of Degree Program
- Class Period/ Attendance Period/ Academic Year・Semester
- Course Registration
- Course Drop
- Attendance/ Grade/ Exam
- Credit Transfer/ Credit Carryover
- Academic Forms
- Education Support
-
Research/Industry
- Research Aims
- Research Findings
- Researcher Search
-
Research Organizations
- UNIST Multi-Interdisciplinary Institute
- IBS Research Groups
-
UNIST Labs
- Department of Mechanical Engineering
- School of Energy and Chemical Engineering
- Department of Civil
- Department of Materials Science and Engineering
- Department of Nuclear Engineering
- Department of Industrial Engineering
- Department of Design
- Department of Biomedical Engineering
- Department of Biological Sciences
- Department of Electrical Engineering
- Department of Computer Science and Engineering
- Department of Mathematical Sciences
- Department of Chemistry
- Department of Physics
- School of Business Administration
- Graduate School of Carbon Neutrality
- Graduate School of Artificial Intelligence
- Research Support
- University-Industry Relations
- Campus Life
- News Center
- About UNIST
-
etc
- UNIST Bulletin
- Work-Life Balance Support System
- UNIST Gender Equality Plan
- Faculty Invitation for Tenure Track
- Faculty Invitation for Non-Tenure Track
- Board Meeting Minutes
- University Council Meeting Minutes
- Administrative Service Charter
- Privacy Policy
- Copyright Policy
- Rejection of Unauthorized Email Collection
- Information Disclosure


Connection Points of Knowledge, Everything About UNIST
Try searching.
Recommended search terms


- portal
- U Academics Innovation Center
- Leadership Center
- Dormitory
- Academic Information Center
- International Students Support
- Browse Open Courses
- Course Registration
- Graduation Requirements for Graduation
- Academic Leave of Absence/ Academic Return
- Military Service
- Certificate Issuance
- Academic Calendar
- Scholarships
- Campus Map
- Campus Life Guidebook
- Health Care Center
- Human Rights Center
- portal
- Job Opening
- Announcement for Bid
- UNIST AI Services
- UNIST Daycare Center
- Sports Center
- UI Downloads
- Announcement
- Recruitment of Professors (Non-tenure)
- Faculty Invitation for Tenure Track
- UNIST Academic Information Center
- Office of Research Facilities and Training
- Office of Research Affairs
- Rule Management System
- Academic Calendar


NEWS CENTER
Discover not only Research Findings and event news, but also the diverse facets of UNIST presented by reporters and writers.
News Center
UNIST News
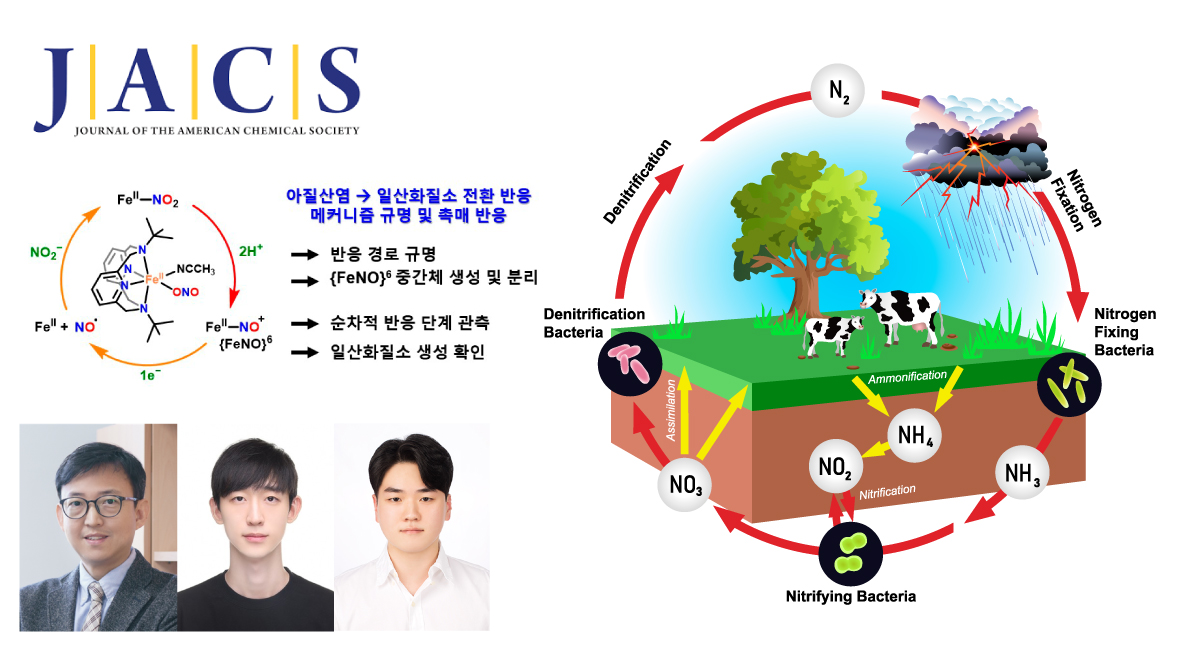
Breakthrough Observation of Transient Intermediate in Nitrite-to-Nitric Oxide Conversion
The findings were published in the Journal of the American Chemical Society (JACS) on March 20, 2026.
- Research
- JooHyeon Heo
- 2026.04.16
- 263
Abstract
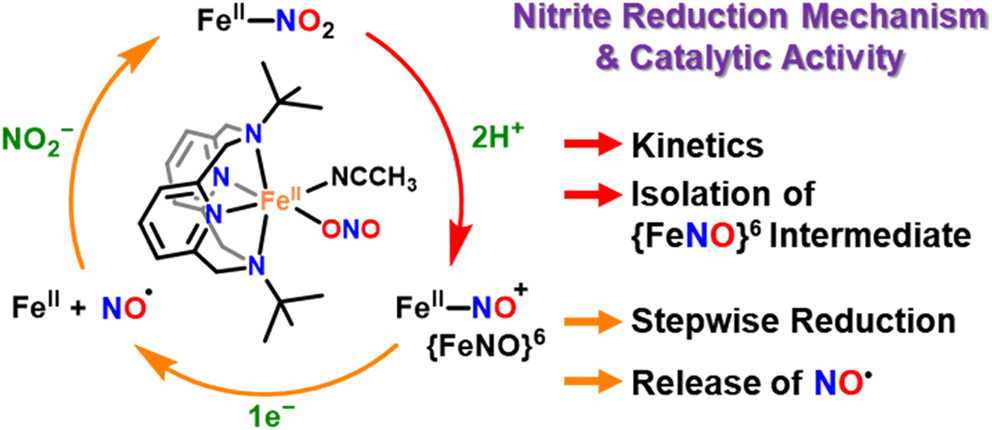
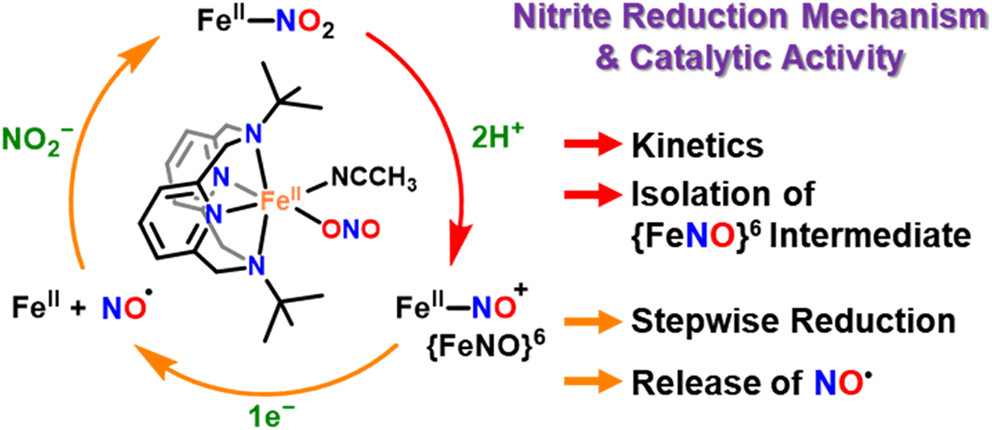
Researchers from UNIST and Jeonbuk National University have, for the first time, captured and analyzed a short-lived iron (Fe)-based intermediate involved in converting nitrite (NO2–) to nitric oxide (NO)—a key process in the nitrogen cycle and biological signaling. This discovery, made at ultra-low temperatures, provides new insights into how vital molecules are produced in nature and in biological systems.
Using a specialized Fe(ll)-nitrite complex and reaction conditions at -40°C, Professor Jaeheung Cho from the Department of Chemistry at UNIST, in collaboration with Professor Kyung-Bin Cho at Jeonbuk National University isolated the elusive {FeNO}⁶ intermediate, a critical step preceding NO release. Spectroscopic and computational analyses confirmed that this species forms after NO2– accepts a proton and undergoes bond cleavage, with the nitrogen-oxygen ion binding to Fe. Further electron transfer then liberates NO.
The study also revealed that the reaction pathway varies depending on whether proton and electron transfers occur sequentially or simultaneously, providing nuanced insight into reaction mechanisms.
Professor Cho remarked, “This is the first direct observation of the intermediate in NO2– reduction to NO. Understanding this step could inform targeted therapies for vascular diseases and inspire the design of new catalysts with improved efficiency.”
According to the research team, this discovery advances fundamental knowledge of nitrogen cycle chemistry and biological NO production, with potential applications in medicine and sustainable catalysis. By elucidating the reaction pathway, the research opens avenues for developing innovative treatments and catalytic systems.
These findings were published in the Journal of the American Chemical Society (JACS) on March 20, 2026. The study has been supported by the Ministry of Science and ICT (MSIT), the National Research Foundation of Korea (NRF), and the Ministry of Health and Welfare (MOHW).
Journal Reference
Seungwon Sun, Youngjin Jeon, Youngseob Lee, et al., “Unveiling an {FeNO}6 Intermediate: A Sequential Mechanistic Investigation of Nitrite Reduction in a Mononuclear Iron(II) Complex,” JACS, (2026).
Related Links